Key Concepts
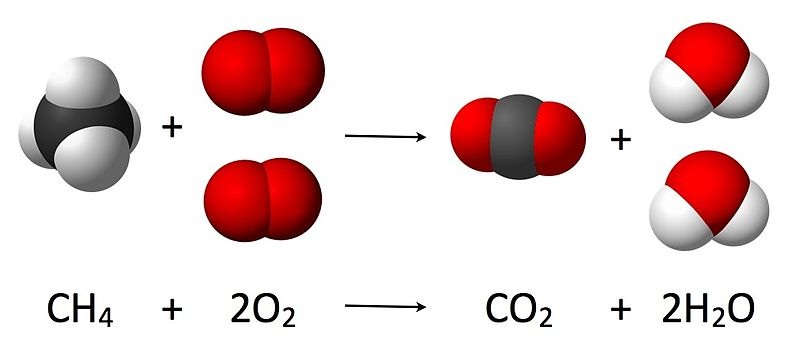
In a chemical equation, the number of molecules of a given substance is represented by a coefficient.
It is also very important to remember that in a chemical equation an arrow takes the place of the word "yields".
In a double replacement reaction, hydrogen chloride and sodium hydroxide react to produce water and a salt (Hint: Salt is Sodium Chloride).
Remember that when writing the symbol for a diatomic element you always needs to have a subscript of 2 after each element's symbol.
Balancing Equations Rules
1. Determine correct formulas and physical states.
2. Write equation with reactants on left and products on right.
3. Count the number of atoms of each element in reactants and products.
4. Balance equation using coefficients. NEVER CHANGE SUBSCRIPTS.
5. Check each atom or poly-atomic ion to be sure equation is balanced.
6. Make sure coefficients are in the lowest possible ratio.
It is also very important to remember that in a chemical equation an arrow takes the place of the word "yields".
In a double replacement reaction, hydrogen chloride and sodium hydroxide react to produce water and a salt (Hint: Salt is Sodium Chloride).
Remember that when writing the symbol for a diatomic element you always needs to have a subscript of 2 after each element's symbol.
Balancing Equations Rules
1. Determine correct formulas and physical states.
2. Write equation with reactants on left and products on right.
3. Count the number of atoms of each element in reactants and products.
4. Balance equation using coefficients. NEVER CHANGE SUBSCRIPTS.
5. Check each atom or poly-atomic ion to be sure equation is balanced.
6. Make sure coefficients are in the lowest possible ratio.
Chemical Reactions - Created with Haiku Deck, presentation software that inspires
"Chemical Equations." YouTube. YouTube, 2 Sept. 2012. Web. 29 May 2014.
"How to Determine Significant Figures and Use Sig Fig Rules - Chemistry."YouTube. YouTube, 7 Nov. 2010. Web. 29 May 2014.
Symbols Used In Chemical Reactions